Rare Earth Elements (REE’s)
Rare earths elements (REEs) are a series of elements found in the earth’s crust that are becoming critical to the future sustainability of many modern technologies including, electric cars, wind turbines, smart phones along with many other applications. While these elements themselves aren’t rare, finding a project with high enough concentrations to make mining economically viable is in fact rare.
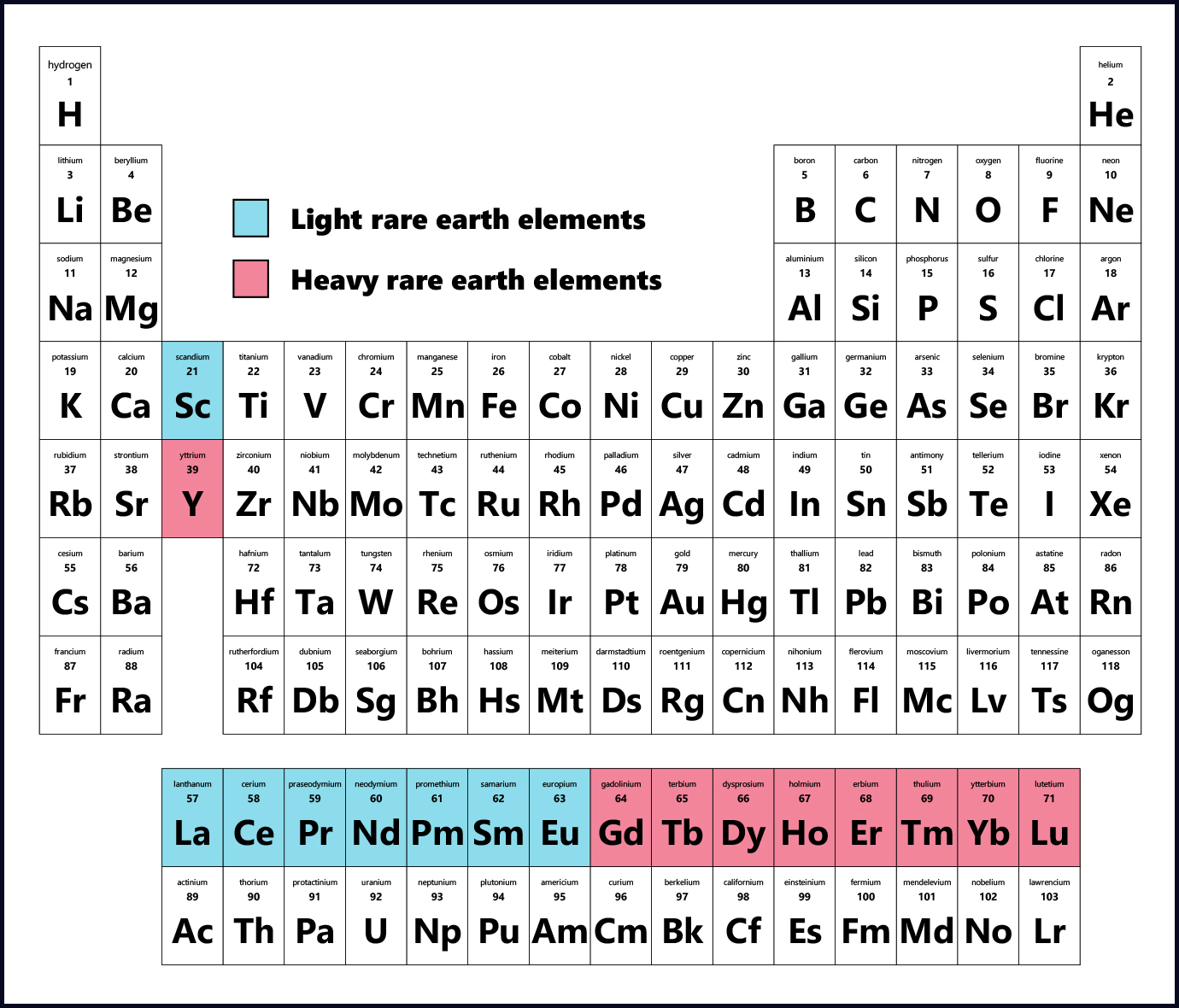
REEs are those in the lanthanide series of the periodic table and include: lanthanum (La), cerium (Ce), praseodymium (Pr), neodymium (Nd), promethium (Pm), samarium (Sm), europium (Eu), gadolinium (Gd), terbium (Tb), dysprosium (Dy), holmium (Ho), erbium (Er), thulium (Tm), ytterbium (Yb), lutetium (Lu) and yttrium (Y). Scandium (Sc) is also commonly considered a rare earth element by the US Geological Survey.